Paper or Plastic?
Intro:
When you and your family go shopping, which do you choose to use? Paper or Plastic? Why do you choose it? Ease of use? Strength or durability? Eco-Friendly? Cost Efficiency? Multiple Uses? Some people prefer paper because paper is known to break down much easier in the environment, meaning it’s biodegradable. However, plastic bags are cheaper to make and uses a lot less energy to make them; which is also a plus for the environment as well. What about other aspects, like carry capacity, tearing, and other forces affecting the durability of the bag. Think about why you use one over the other, then ask yourself, which is better - paper or plastic?
When you and your family go shopping, which do you choose to use? Paper or Plastic? Why do you choose it? Ease of use? Strength or durability? Eco-Friendly? Cost Efficiency? Multiple Uses? Some people prefer paper because paper is known to break down much easier in the environment, meaning it’s biodegradable. However, plastic bags are cheaper to make and uses a lot less energy to make them; which is also a plus for the environment as well. What about other aspects, like carry capacity, tearing, and other forces affecting the durability of the bag. Think about why you use one over the other, then ask yourself, which is better - paper or plastic?
Question:
Materials:
Background Info:
- Which is better, paper or plastic?
- Which would you choose to carry these different items?
- Canned Goods
- 3 Heavy Boxes of Minute Rice
- 2 Gallons of Milk
- 1 lb of Nails
Materials:
- 3 to 4 Different Bags
- Brown Paper Bag (P)
- Plastic Grocery Bag (G)
- Husky Trash Bag (H)
- Black Trash Bag (B)
- 6 (1 in x 12 in) strips of each bag material.
- D Rings
- 20 & 50 Newton Force Meters
Background Info:
- A Force is a push or a pull.
- It is measured in Newtons [Metric = N, Standard = Ft.lbs]
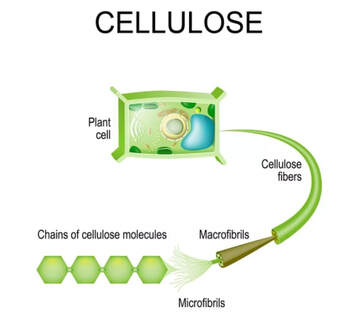
- Paper bags are made from cellulose, which is the cell wall of plant material.
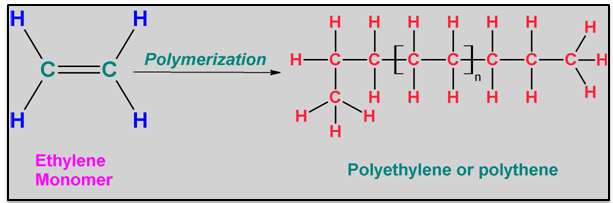
- Plastic Bags are made of Polyethylene from the Ethylene Monomer
- There are 3 types of Polyethylene
- High Density Polyethylene (HDPE)
- Low Density Polyehthylene (LDPE)
- Linear Low Density Polyethylene (LLDPE). This is the one that plastic bags are made of.
- There are 3 types of Polyethylene
Polyethylene
|
Physical Properties:
The melting point and glass transition temperature for the polyethylene are dependent on the type of plastics they belong to. For common commercial grades of medium- and high-density polyethylene, the melting point is typically in the range of 120 to 180 °C. The melting point for average, commercial, low-density polyethylene is typically 105 to 115 °C. These temperatures vary strongly with the type of polyethylene. Polyethylene is of low strength, hardness, and rigidity, but has high ductility and impact strength as well as low friction. It shows strong creep under persistent force, which can be reduced by the addition of short fibres. It feels waxy when touched. |
Chemical Properties:
Polyethylene consists of nonpolar, saturated, high molecular weight hydrocarbons. Therefore, its chemical behaviour is similar to paraffin. Most LDPE, MDPE, and HDPE grades have excellent chemical resistance, meaning they can not be attacked by strong acids or strong bases, and are resistant to gentle oxidants and reducing agents. Polyethylene (other than cross-linked polyethylene) usually dissolved at elevated temperatures in aromatic hydrocarbons such as toluene or xylene, or in chlorinated solvents such as trichloroethane or trichlorobenzene. Polyethylene absorbs almost no water. Polyethylene burns slowly with a blue flame having a yellow tip and gives off an odour of paraffin (similar to candle flame). The material continues burning on the removal of the flame source and produces a drip. |
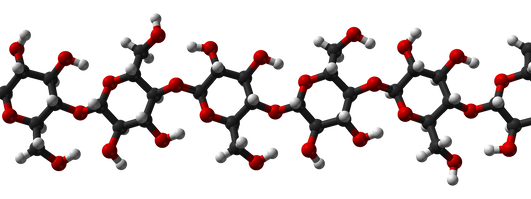
Cellulose
Cellulose is an organic compound with the formula (C6H10O5)n, a polysaccharide consisting of a linear chain of several hundred to many thousands of β(1→4) linked D-glucose units.[3][4] Cellulose is an important structural component of the primary cell wall of green plants, many forms of algae and the oomycetes. Some species of bacteria secrete it to form biofilms.[5] Cellulose is the most abundant organic polymer on Earth.[6] The cellulose content of cotton fiber is 90%, that of wood is 40–50%, and that of dried hemp is approximately 57%.[7][8][9]
Cellulose is mainly used to produce paperboard and paper. Smaller quantities are converted into a wide variety of derivative products such as cellophane and rayon. Conversion of cellulose from energy crops into biofuels such as cellulosic ethanol is under development as a renewable fuel source. Cellulose for industrial use is mainly obtained from wood pulp and cotton.[6]
Cellulose is mainly used to produce paperboard and paper. Smaller quantities are converted into a wide variety of derivative products such as cellophane and rayon. Conversion of cellulose from energy crops into biofuels such as cellulosic ethanol is under development as a renewable fuel source. Cellulose for industrial use is mainly obtained from wood pulp and cotton.[6]
Professional Materials Testing (Engineering)
Tests:
- Pull Test: Place a D-Ring in the middle of the strip and pull downwards.
- Rip Test: Punch a hole in the middle of a strip with a force meter, then tear sideways.
- Tear Test: Literally tear the material in half and get a feel for how easy it is to get the tearing started and how it feels to keep tearing.
- Poke Test: Take a pencil and poke it through the material.
- Open Test: Have an idea of what test you want to run? Go for it!